Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. The value of the energies of photons does depend on the available energy levels in the atom. Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. Here is my answer, but I would encourage you to explore this and similar questions further. It successfully explains that electrons move in permitted orbit with fixed energy and angular momentum.Its a really good question. It doesn’t explain the orbits or energy level. It is based on the atomic spectra of the Hydrogen atom. 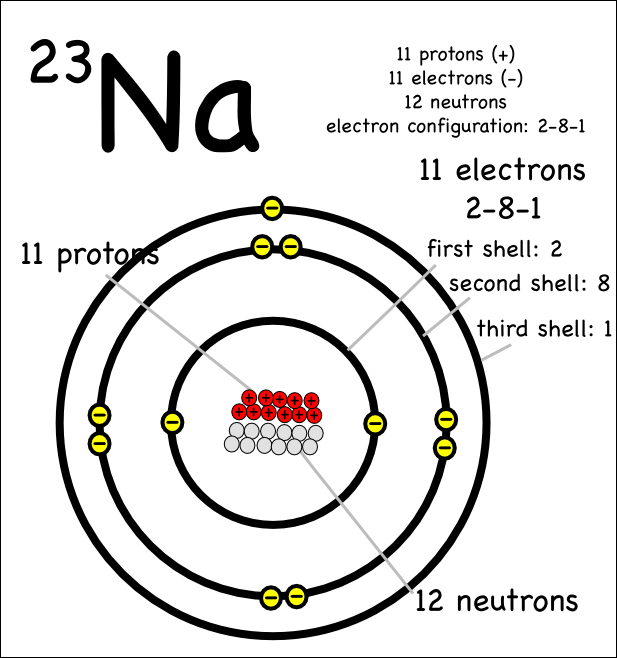
It is based on an alpha particle scattering experiment. It accounts for the stability of an atom. 
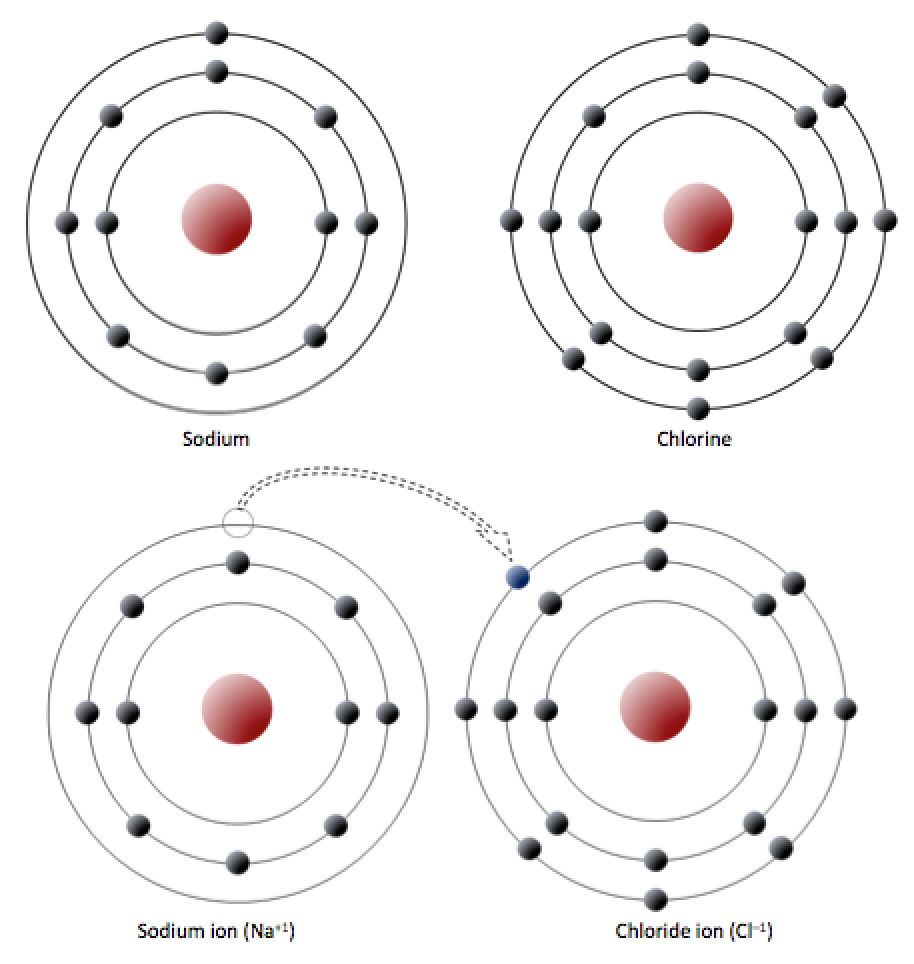
It couldn’t explain the stability of the atom. It states that the nucleus is positively charged and is surrounded by electrons (negatively charged electronsĪccording to Bohr’s atomic model, “a positively charged nucleus is surrounded by electrons moving in fixed orbits.”
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
